
Influenza Vaccination During Pregnancy: A Very Bad Idea
June 2006
“Influenza vaccination during all trimesters of pregnancy is now universally recommended in the United States. We critically reviewed the influenza vaccination policy of the CDC’s Advisory Committee on Immunization Practice (ACIP) and the citations that were used to support their recommendations.” Thus began the abstract to a paper by David Ayoub, MD, and me, which was just published in the summer issue of the Journal of American Physicians and Surgeons.
It continued, “The ACIP’s citations and the current literature indicate that influenza infection is rarely a threat to a normal pregnancy. There is no convincing evidence of the effectiveness of influenza vaccination during this critical period. No studies have adequately assessed the risk of influenza vaccination during pregnancy and animal safety testing is lacking. Thimerosal, a mercury based preservative present in most inactivated formulations of the vaccine has been implicated in human neurodevelopmental disorders, including autism, and a broad range of animal and experimental reproductive toxicities including teratogenicity, mutagenicity and fetal death. Thimerosal is classified as a human teratogen.
“Conclusions: The ACIP policy recommendation of routinely administering influenza vaccine during pregnancy is ill advised, unsupported by current scientific literature and should be withdrawn. Use of thimerosal during pregnancy should be contraindicated.”
This major review — “Influenza Vaccination during Pregnancy: A Critical Assessment of the Recommendations of the Advisory Committee on Immunization Practices” — is the first and only review of the subject that was not financed by the vaccine manufacturers or instigated by the Centers for Disease Control and Prevention (CDC) to justify its recommendation to inject pregnant women with a vaccine of unproven efficacy and safety and that, to date, still contains thimerosal, a mercury preservative.
Recently, I had the pleasure of co-writing with Ayoub, “The Battle Of The States: What Happened In Illinois?” In that report, we exposed the incredible shenanigans that went on in Illinois, when a law banning the use of thimerosal in vaccines was passed almost unanimously — one dissenting vote — by both Houses of the Legislature only to be cleverly circumvented by a joint effort of the State Department of Health, the Illinois Chapter of the American Academy of Pediatrics and the CDC.
Our Journal of American Physicians and Surgeons article critically examines all information available to the Advisory Committee on Immunization Practices (ACIP) and all other data available in the medical press worldwide. It also cites 60 references.
The following represents a very limited review of reports that were filed with the Vaccine Adverse Events Reporting System (VAERS), following influenza vaccination during pregnancy.
It should be pointed out that the CDC and the Food and Drug Administration (FDA), who created VAERS, like to stress that filing a report with VAERS does not necessarily mean the administered vaccine actually caused the adverse event reported.
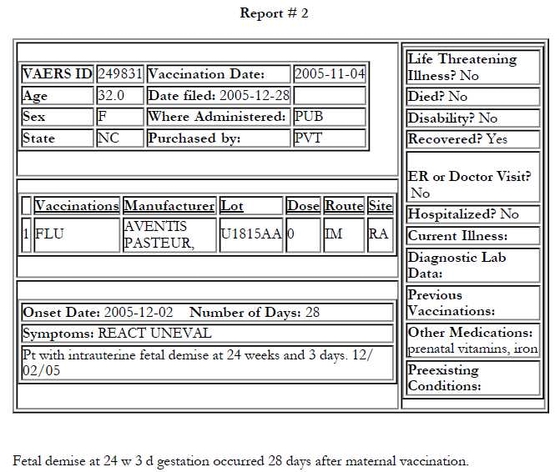
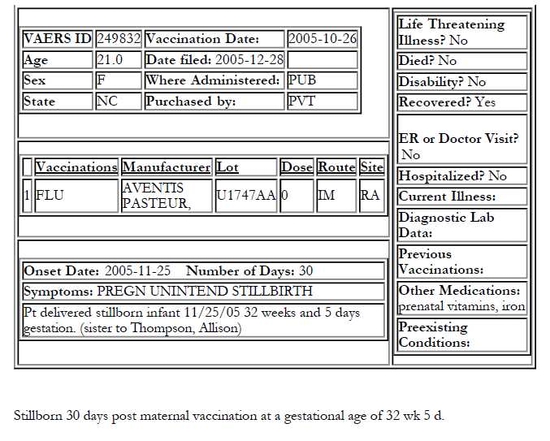
The following four VAERS reports — all filed on December 28, 2005 — may suggest therwise. All four patients were from North Carolina, all received the same brand of injectable influenza vaccine and two received doses from the same lot.

Report 3

To review:
All four North Carolina cases were reported to VAERS on Dec. 28, 2005
The mother in the first report was vaccinated on Oct. 25, 2005
The three other mothers were vaccinated on Oct. 26, 2005
They all received Fluzone by Sanofi Pasteur
Case No. 1 and No. 3 received vaccines from the same lot
All fetal deaths occurred within three to four weeks of maternal vaccination
Two of the mothers may have been sisters
Another unclear and rather confusing report submitted on Jan. 1, 2006 suggests that there actually could have been a fifth incident of fetal demise somehow connected to the above four cases.
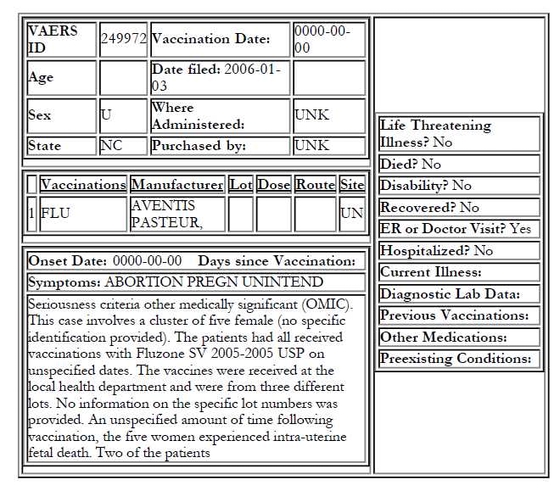
Note:
The tone of the report suggests that it was called in by a health professional
A “cluster” of five females is mentioned
The women received the same brand of influenza vaccine
The administered vaccines came from only three vaccine lots
The vaccines were administered at/by the local health department
At least four reports were filed with VAERS on the same day
All of these facts did not seem to ring any bells or to raise any eyebrows either at the “local health department” in North Carolina or at the VAERS intake office where they were received.
One can only imagine the commotion if there had been five cases of measles or hepatitis A diagnosed at the same North Carolina local health department and reported simultaneously. Sirens would have sounded, the CDC would have landed field investigators and thousands of doses of vaccine (or immune globulin) would have been airlifted. There would have been news reports around the clock and, in the case of hepatitis A, a fast food restaurant would have been closed for a week and scoured thoroughly by people in hazmat suits.
And, yet, we had five incidents of viable fetuses dying and nothing was done and not much was said.
Thanks to Red Flags, the ACIP and Sanofi Pasteur have now been informed.
* * * *
Last Updated: Sun, 2006-06-04 12:27
Copyright ©2006. It is prohibited by copyright law to quote, in whole or in part, from any article on this site, without the expressed consent of the author of the article. Links, however, can be made to articles on this site – You need a Web standards compliant browser to view this site properly
It is likely that the above reports were only filed because the events were indeed “clustered” and the women were vaccinated at a “local health department.” It is just as likely that many isolated similar situations are never reported.
It is also almost certain that an overwhelming number of early miscarriages following influenza vaccination are never reported.On Dec. 15, 2005, a 39-year-old mother from Mississippi contacted VAERS and filed report 249306. In it, she stated that she received a dose of influenza vaccine on Nov. 29, 2005 and that on Dec. 9, an ultrasound revealed the absence of a fetal heartbeat.
Another report (# 231630), filed on Dec. 28, 2004, revealed that a 31-year-old Minnesota woman, who was six weeks pregnant, received a flu vaccination on Oct. 28 and miscarried one week later.
In our Journal of American Physicians and Surgeons study, we also mention the real risk that in a busy practice, the intranasal live influenza vaccine could be administered, by error, to a pregnant woman. The vaccine is absolutely contraindicated during any stage of gestation.
The following is the most recently filed VAERS report under flu vaccination and pregnancy. It confirms our worst fears.
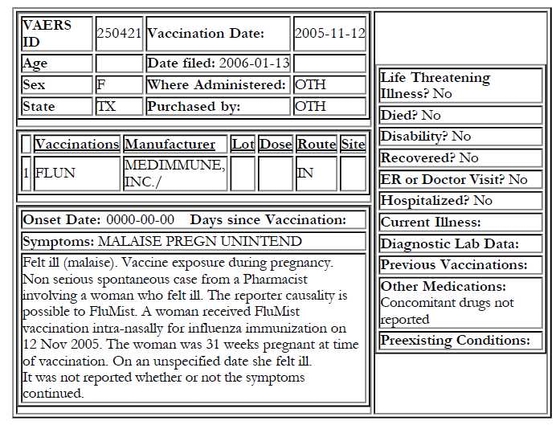
The stilted style and the careful phrasing leave no doubt that the note was composed and written by the VAERS intake clerk.
Logically, one would have expected the clerk who entered the data to ask the reporting pharmacist to have the patient or the doctor call as soon as possible to give more details.
Evidently and most unfortunately, that did not happen. The above was not the only report concerning the live virus vaccine FluMist being administered to a pregnant woman. VAERS report 219257, filed on April 19, 2004, related a similar situation. That patient developed fever, headache, runny nose and sore throat shortly after vaccination.
As we concluded in our Journal of American Physicians and Surgeons report: “Pregnancy should continue to be a time when doctors are highly protective of their patients with regards to any fetal exposure.”
Last Updated: Sun, 2006-06-04 12:27
Copyright ©2006. It is prohibited by copyright law to quote, in whole or in part, from any article on this site, without the expressed consent of the author of the article. Links, however, can be made to articles on this site – You need a Web standards compliant browser to view this site properly