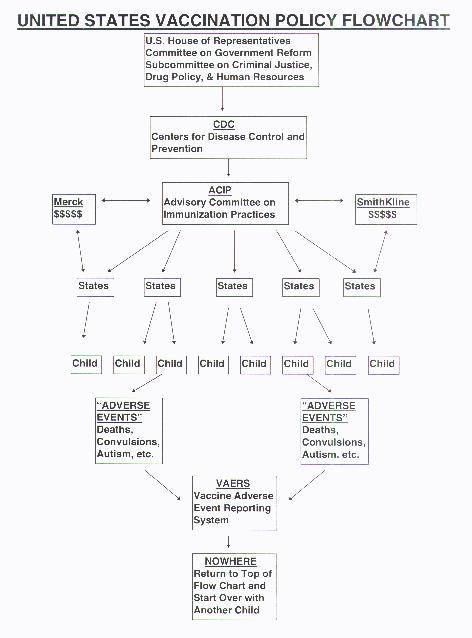
VAERS (Vaccine Adverse Event Reporting System)
http://www.fda.gov/cber/vaers/vaers.htm
[Multiply these by 40 to 100
times to get somewhere near the true figure (see).]
See:
Hiding vaccine reactions (coincidence)
Hiding adverse
reactions (don't report)


[pdf 2016 Summer] Combining Childhood Vaccines at One Visit Is Not Safe Neil Z.
Miller
Miller,
Neil
[vid] Dr Anthony Morris (VAERS)
Morris, Dr Anthony J.
[2010] Nurse experience at call center for
pharmaceutical companies
[2015] A Nurse's Story ~ "I have seen the cover up." For all the
cases I've seen, I have NEVER seen any medical provider report them to VAERS. I
have filed VAERS reports. But I am the ONLY nurse I have EVER met that files
VAERS reports. I also have NEVER met a doctor that filed a VAERS report.....The
number one place parents bring their kids in the event of a vaccine reaction is
the E.R., and as an E.R. staffer, I have NEVER met anyone who filed one, in
spite of seeing hundreds of cases of obvious vaccine associated harm come
through. What does that say about reported numbers? The CDC/HHS admits that
VAERS is under-reported, and probably only representative of 1/10th the actual
number of injuries. I contest that, and from personal experience, I would say
the numbers in VAERS are more like 1/1000th the actual numbers, not 1/10th.
[2011 Oct] 61% of Vaccine Providers Don�t Know What
a Vaccine Adverse Reaction Is If
you want to entrust yourself and your children to this Three Blind
Mice (see no evil, hear no evil, speak no evil) vaccine system �
that is certainly your right, but if something goes wrong don�t expect any
recognition, sympathy or cure from the medical system. You�ll end up being a
statistic in the FDA VAERS system and it will be labeled a coincidence.......Even
if vaccine providers on the front lines of sticking needles in people were to
cause, observe and report a vaccine adverse reaction (such as described above)
to the FDA Adverse Event Reporting System (VAERS) absolutely nothing would
happen. The FDA collects those reports, puts them in a drawer and forgets about
them. Here is the current body count in VAERs: Hepatitis B vaccine 50,275
reports � 979 deaths, DTaP vaccine (pertussis, diptheria, tetanus) 50,345
reports � 785 deaths, MMR vaccine 58,887 reports � 300 deaths, Gardasil vaccine
22,563 reports � 99 deaths.
[2011 May] Six Times More Measles Vaccine Adverse
Reaction Reports Than Measles Cases in 2011
there have been 698 FDA Vaccine Adverse Event Reporting System (VAERS) reports
related to MMR, MMRV (MMR plus varicella) and measles vaccines in 2011 �
including 4 deaths and 280 emergency room visits. 698 VAERS reports are
almost six times more than the number of measles cases
[2011 Jan] VAERS
- I�m Not Laughing By Sandy Gottstein
[2010] VAERS
- Is the Joke On Us by Sandy Gottstein
There have so far been almost 301,000 vaccine-associated adverse reactions
reported to VAERS. Even without the fact of under-reporting
in a passive reporting system being taken into account (thought to be
between 1-2%
even for serious events), that�s a lot of reactions. With
the correction, it amounts to the possibility that the number reported could
represent as many as 30,100,000 adverse vaccine-associated events.
[VAERS 1983-2008] 357 events with Symptom is Abortion spontaneous
[VAERS] Found 25 events with Vaccine
as FLU and Symptom is Abortion
[Aug 2004]
CDC AIMS TO DENY
OUTSIDE ACCESS TO VACCINE DATABASE.
VAERS-217 Deaths from Prevnar
since May2000
https://secure.vaers.org/VaersDataEntryintro.htm
http://users.adelphia.net/~cdc/VAERS.htm#CSonVAERS
Adverse Reactions reported to VAERS from 1999-2002, age
0-6 years.
Vaccine Safety Datalink
VAERS reports extracted by Sandy Mintz http://www.vaccinationnews.com/
VAERS database 1990-2000 made available by
Steven
Rubin
http://www.std.org/strubin/VAERSDB.zip
whale.to/zip/VAERSDB.zip
http://www.whale.to/zip/VAERSDB.zip
VAERS database 1990 (Excel 582kb)
VAERS database deaths 1990 (Excel)
How to use database
http://www.nccn.net/~wwithin/vaers.htm
VAERS database (not on internet)
Source: whale/zip/vaers.mdb
MICHAEL BELKIN'S WRITTEN TESTIMONY
TO CONGRESS
difference between VAERS AND VICP
[Lies]
Quackwatch vaccine information (vaccine deaths)
Braun
MM, Mootrey GT, Salive ME, Chen RT, Ellenberg SS. Infant
immunization with acellular pertussis vaccines in the United States: assessment of the
first two years' data from the Vaccine Adverse Event Reporting System (VAERS). Pediatrics. 2000 Oct;106(4):E51. PMID: 11015546 [PubMed - indexed for MEDLINE]
OBJECTIVE: To evaluate the safety of infant immunization with acellular
pertussis vaccines in the United States. BACKGROUND: The US Food and Drug Administration
approved the first acellular pertussis vaccine for use in infants in the United States on
July 31, 1996. OUTCOME MEASURES: Adverse events in the United States after infant
immunization with pertussis-containing vaccines, representing temporal (but not
necessarily causal) associations between vaccinations and adverse events. DATA SOURCE:
Reports to the Vaccine Adverse Event Reporting System (VAERS), a passive national
surveillance system. DESIGN: Reports concerning infant immunization against pertussis
between January 1, 1995 (when whole-cell vaccine was in exclusive use)
and June 30, 1998 (when acellular vaccine was in predominant use) were
analyzed, if the reports were entered into the VAERS database by November 30, 1998.
RESULTS: During the study, there were 285 reports involving death, 971 nonfatal
serious reports, and 4514 less serious reports after immunization with any
pertussis-containing vaccine. For 1995 there were 2071 reports; in 1996 there
were 1894 reports; in 1997 there were 1314 reports, and in the first half of 1998 there
were 491 reports. Diphtheria-tetanus-pertussis vaccine (DTP) was cited in 1939 reports,
diphtheria-tetanus-whole-cell pertussis-Haemophilus influenzae type b vaccine (DTPH) in
2918 reports, and diphtheria-tetanus-acellular pertussis vaccine (DTaP) in 913 reports.
The annual number of deaths during the study was 85 in 1995, 82 in 1996, 77 in 1997, and
41 in the first half of 1998. The annual number of reported events categorized as nonfatal
serious (defined as events involving initial hospitalization, prolongation of
hospitalization, life-threatening illness, or permanent disability) to VAERS for all
pertussis-containing vaccines declined: 334 in 1995, 311 in 1996, 233 in 1997, and 93 in
the first half of 1998. Similarly, the annual number of less serious reports to VAERS for
pertussis-containing vaccines declined: 1652 in 1995, 1501 in 1996, 1004 in 1997, and 357
in the first half of 1998. A comparison of the adverse event profiles (proportional
distributions) for DTaP, DTP, and DTPH, as well as an analysis of specific adverse events
considered in a 1991 Institute of Medicine report on the safety of
diphtheria-tetanus-pertussis vaccine, did not identify any new, clear safety concerns.
CONCLUSIONS: These findings reflect the administration of millions of doses of acellular
pertussis vaccine and are reassuring with regard to the safety of marketed acellular
pertussis vaccines. VAERS data, although subject to the limitations of passive
surveillance, support the prelicensure data with regard to the safety of the US-licensed
acellular pertussis vaccines that we evaluated.